
Monday, May 9, 2016
Airbag stuff...
I managed to pass the lab today, I know, what a surprise! I was super nervous for it, but I thankfully came through! I think everyone in my class passed the lab as well, so that's cool.


Probably got an A?
On Thursday, we took a quiz on gas laws. The quiz was not that bad at all, and I think I scored pretty well. The lectures we've learned so far were not too hard and only consisted of formulas. I am looking forward to our next lessons.
| http://www.gettyimages.com/detail/photo/whew-what-a-relief-royalty-free-image/471428407 |
Something with pvnrt
Today, we had another lecture for the gas laws unit. This was yet another formula lecture, one that was not difficult at all and mostly involved basic plug-n-chug. I am really nervous for the lab on Monday as it is pass-fail, and I don't 100% trust myself with being accurate in my calculations. Hopefully, it goes well.
Some review links:
https://www.khanacademy.org/science/chemistry/gases-and-kinetic-molecular-theory/ideal-gas-laws/v/ideal-gas-equation-pv-nrt
http://www.learnchem.net/practice/gases.shtml
http://www.mikeblaber.org/oldwine/chm1045/notes/Gases/IdealGas/Gases04.htm
| http://www.calctool.org/CALC/chem/c_thermo/ideal_gas |
Some review links:
https://www.khanacademy.org/science/chemistry/gases-and-kinetic-molecular-theory/ideal-gas-laws/v/ideal-gas-equation-pv-nrt
http://www.learnchem.net/practice/gases.shtml
http://www.mikeblaber.org/oldwine/chm1045/notes/Gases/IdealGas/Gases04.htm
Gas Lectures
The gas laws unit has proven to be pretty straightforward and simple. The first lectures we've had are centered on one major formula: the combined gas law formula. We've gone through various different problems concerning the gas law, only with different constants. Additionally, we've learned different units of pressure, which I believe is the only difficult part of this unit: memorizing all the different conversions and units.
Some links I've been using to study with for the quiz tomorrow:
| https://prezi.com/8ywewmiga8tq/combined-gas-law/ |
Some links I've been using to study with for the quiz tomorrow:
since when does it take 15 min to complete a test
The unit test for energy was a bit of a doozy. Since our schedule for the day was messed up, we were only given 28 minutes to complete the test. Seems impossible, right? It actually wasn't as bad as I thought it was going to be. There were only 20 questions, and I managed to finish with a couple minutes to spare. We got our grades back and I managed to score what I needed to keep my grade where it needs to be in order to exempt the final exam.
Studying for the final
| https://commons.wikimedia.org/wiki/File:Phase_changes.svg |
| http://sonoranweeklyreview.com/with-linn-energy-llc-nasdaqline-sandridge-energy-sdoc-and-midstates-petroleum-mpoy-depleting-their-revolving-credit-lines-banks-scramble/ |
Some links I used for studying:
http://www.aboutthemcat.org/chemistry/thermochemistry.php
https://www.chem.wisc.edu/deptfiles/genchem/netorial/rottosen/tutorial/modules/intermolecular_forces/02imf/imf2.htm
https://www.sophia.org/tutorials/thermochemistry-calculating-energy-of-phase-change
https://www.khanacademy.org/test-prep/mcat/chemical-processes/covalent-bonds/a/intramolecular-and-intermolecular-forces
Metal, Heat, some other stuff...
Yesterday, we did a lab to measure the specific heats of copper and lead by heating the metals in a hot water bath and figuring out the variables for MCAT. I'm pretty satisfied because our copper trial yielded only a 4% error for the specific heat!
Some review links for the first lesson:
Boat Races...
Last week, we conducted our boat races. I'm proud to say that my partner and I won last place. It was a really fun experience, and it definitely was a relief from the usual lecture/test stress. We made it out of an eggnog carton...

A real boat that runs on biodiesel: https://www.youtube.com/watch?v=4EggCi3qM2A
So, The biodiesel worked lol
On Friday, we found our biodiesel separated into three layers! The top and bottom layers were impurities, and our actual biodiesel was the middle layer. To test if our biodiesel worked, we filled an empty tea light wick with the biodiesel and lit it. Sure enough, the flame was sustained for quite a bit, indicating that our biodiesel worked great!
Biodiesel, I think?
Here's some research I accumulated regarding the topic of Biodiesel:
General Applications
Benefits;
General Applications
- Biodiesel can be used in pure form (B100) or may be blended with petroleum diesel at any concentration in most injection pump diesel engines
- With 80-90% of oil spill costs invested in shoreline cleanup, there is a search for more efficient and cost-effective methods to extract oil spills from the shorelines. Biodiesel has displayed its capacity to significantly dissolve crude oil, depending on the source of the fatty acids
- Depending on the engine, this might include high pressure injection pumps, pump injectors (also called unit injectors) and fuel injectors
Benefits;
- domestically produced, clean-burning, renewable substitute for petroleum diesel. Using biodiesel as a vehicle fuel increases energy security, improves air quality and the environment, and provides safety benefits
- Biodiesel is produced in the U.S. and used in conventional diesel engines, directly substituting for or extending supplies of traditional petroleum diesel-- no need for petroleum imports
- reduces nitrogen oxide (NOx) emissions to near zero levels
- reduces greenhouse gas emissions because carbon dioxide released from biodiesel combustion is offset by the carbon dioxide absorbed while growing the soybeans or other feedstock
- B100 use reduces carbon dioxide emissions by more than 75% compared with petroleum diesel. Using B20 reduces carbon dioxide emissions by 15%
- Biodiesel improves fuel lubricity and raises the cetane number of the fuel
- Diesel engines depend on the lubricity of the fuel to keep moving parts from wearing
How to make biodiesel
On Thursday, we began the process of making our own biodiesel! First, we took recycled peanut oil and combined it with a mixture of KOH and methanol. Then, we put a flask of this mixture into a hot water bath and boiled it for about 20 minutes. Lastly, we let it cool and settle, upon which it began to separate into two layers.
Make your own biodiesel at home!: http://www.make-biodiesel.org/
 |
| Hot water bath |
 |
| Viscosity test |
Make your own biodiesel at home!: http://www.make-biodiesel.org/
How it's made: https://www.youtube.com/watch?v=xLa83KIaEyw
Bio Diesel Video
This past week, we've been working on a video project to submit to the National Lung Association biodiesel contest. Our video explained the benefits and debunked myths about biodiesel. We made a stop-motion video which took quite a while as we had to take multiple pictures to create a full-length animated video. However, it was definitely worth it as our final product was really cool! I'm really looking forward to checking out other students' videos.
Unit Exam, not too bad
Our unit exam for the chemical bonding unit was yesterday. Initially, I was nervous about the content of the unit as a whole, it was a little tricky to study and remember, but I received a perfectly adequate grade. It was Lewis structure heavy, along with some emphasis on electronic geometry & molecular shape. Here are some fun pictures pertaining to our unit.


More lectures...
Chemical Bonding Lectures
Our current unit is based on a single concept: a (covalent) bond is formed by sharing electrons. Atoms are constantly in search of electrons to fill their valence shells, which will make them stable. More often than not, it's 8 electrons except for Be, B, H, & He. We learned lewis structures, formal charge, electron geometry, and much more involving bonding.
Modeling Activity
Last week, we went to the library for a lab. It was interesting as it was in the library where we drew on the tables and played around with building pieces to create Lewis structures (felt just like kindergarden again). I enjoyed this lab as it allowed me to practice Lewis strucutres, something I am still quite uneasy about. It was also great to have Mrs. Frankenberg hovering around, available to answer any questions (which I had a lot of). I will definitely be finishing the rest of the lab on my own for additional practice.
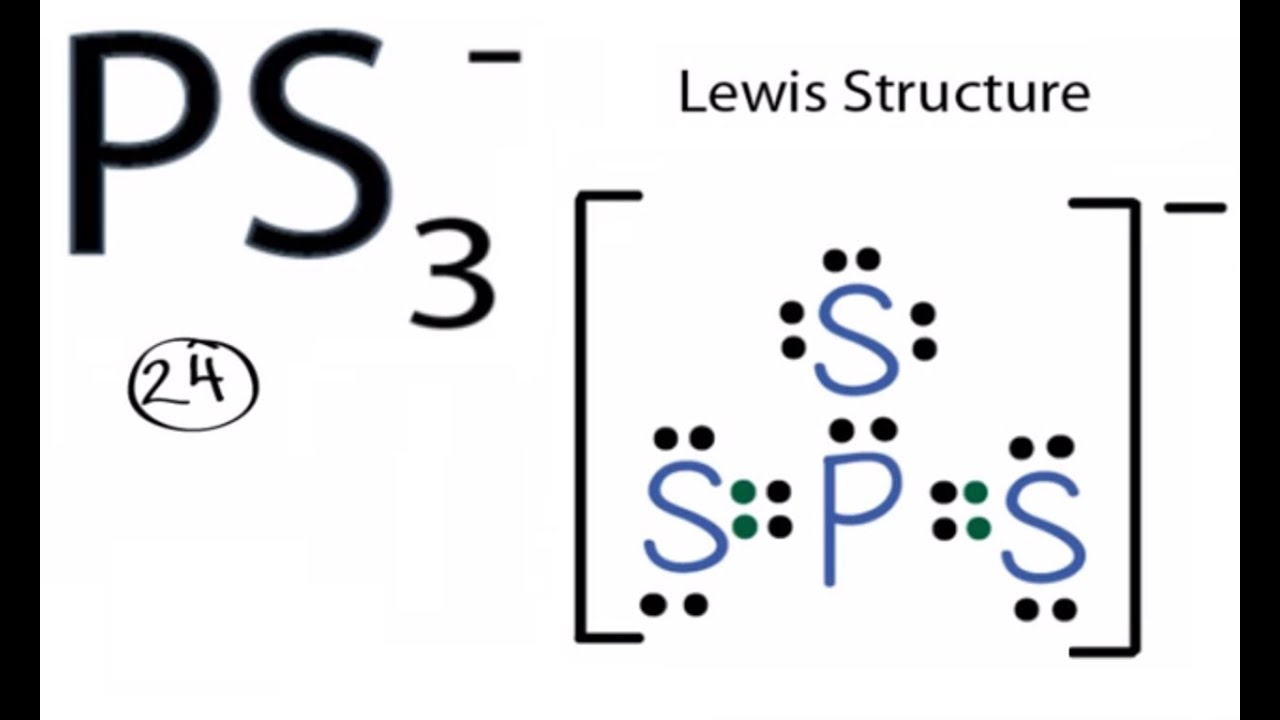
Some more practice: http://web.gccaz.edu/~ksmith8/chm151_files/Lewispractice.pdf
The first day...
Lecture Reflections
We've started the chemical bonding unit, and it's been a bit of a rollercoaster. At first, the concepts were fairly easy to understand, but as the lectures progress, the confusion keeps piling on. I do not believe this unit will be too difficult; it's just the fact that there are SO many things to remember to do problems correctly. I think with a lot of practice and reviewing this unit will not be too bad.
Some practice:
| http://www.slideshare.net/Drahmadfahmi/04-chemical-bonds |
| http://www.roymech.co.uk/Related/Chemistry/Lewis_dot_structure.html |
Some practice:
Saturday, March 5, 2016
Unit Exam
Friday's test went pretty well, I do say. It might've been a smidgen more difficult than the quiz as I was a bit uneasy on a couple questions. Some questions asked about acidity and alkalinity, which I didn't really know how to answer, so I guessed on those. Other than that, the majority of the test was a no-brainer. I feel great on how I did.
Feels good knowing the chemistry behind these beautiful fireworks
Overall, this unit has probably been one of my favorites as the concepts have not been difficult and they are quite interesting to study. I actually enjoyed doing electron configuration problems.
Thursday, March 3, 2016
Periodic Trends Lecture
The final installment of the electronic structure unit is periodic trends. The periodic table follows a couple of trends that can provide details about various atoms (only S and P block atoms though). The different trends were a bit hard to differentiate and remember what each one specifies, but I think with some more studying, they will be easily remembered. I'm feeling pretty good about the test on Friday, but I still definitely need to look over this lecture, which I believe may be the hardest lesson of the unit (but still not that difficult).
https://www.youtube.com/watch?v=XK-WTYncldA
 |
| https://en.wikipedia.org/wiki/Periodic_trends |
https://www.youtube.com/watch?v=XK-WTYncldA
Saturday, February 27, 2016
Weekly Quiz
Yesterday was this unit's weekly quiz. I've felt pretty good about this unit, and that feeling also applied during the quiz. It went by in a breeze, and I had time to double check all of my answers (I basically was able to work out all the questions a second time just to make sure!). It's always a great feeling when a quiz/test doesn't give you a tough time. Also, much of the quiz came directly from the online practice which I always use to study, so that really helped! All in all, I'm feeling great about how I did on the quiz.
Here are some links for future studying:
Tuesday, February 23, 2016
Spectroscopic Analysis Lab
On Tuesday, we performed yet another lab. This lab was not nearly as exciting as the flame test lab. For this lab, we adjusted various knobs on a spectrophotometer and found the amount of light in blue and pink solutions. It was really difficult to adjust the knob so that the number read exactly what it was supposed to. This was extremely time-consuming as we had to re-adjust the knobs for every single different wavelength. Additionally, it was dull as all the lab consisted of was a) adjust the knobs and b) record the number. Not very exciting. But we did get to learn how to make graphs in excel, which isn't as hard as I thought it would be, and I love anything that has to do with computer programs, so that was fun.
Friday, February 19, 2016
Day 1: Lecture
We've started a new unit, and it has been going pretty well. At first, the unit seemed a bit confusing, but it all began to make sense gradually. The first lesson of the unit was on wavelengths of light. We learned the formula for the speed of light (C= frequency x wavelength) and for energy (E= Planck's constant x frequency). These two formulas were easy to understand as problems associated with the lesson are pretty basic plug-and-chug. Yesterday, we learned about the electron configuration of elements. This lesson was chockful of vocab and steps to memorize. It was definitely a bit confusing and time-consuming at first to figure out each element's different electron structure and diagram and to memorize all the little rules for different types of elements. However, I soon began to understand it, and I even think it's a bit fun (Something I have NEVER said about chemistry, so that's saying a lot!).
Some links for review on these lessons:
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch6/electronconfigpractice.html
http://www.sciencegeek.net/Chemistry/taters/Unit2ElectronNotations.htm
https://www.youtube.com/watch?v=2AFPfg0Como
| http://chemwiki.ucdavis.edu/Core/Inorganic_Chemistry/Electronic_Structure_of_Atoms_and_Molecules/Electronic_Configurations |
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch6/electronconfigpractice.html
http://www.sciencegeek.net/Chemistry/taters/Unit2ElectronNotations.htm
https://www.youtube.com/watch?v=2AFPfg0Como
On the other hand, our acid-base test scores have arrived, and I am incredibly happy about my score. I did a lot better than I expected, and being that that unit was supposedly the hardest unit of the year, I feel really great. Thank the chemistry gods.
Thursday, February 18, 2016
Flame Test Lab
Today in class we had another lab, one which may be my favorite lab we've done all year: the Flame Test Lab. Basically, we placed several different chemical compounds and solutions in the flame of a Bunsen burner to see the various colors each one produced. It was really awesome to see the vivid colors that varied among each metal. The smell was really reminiscent of summer (fireworks, bonfire, you get the picture) when we first walked in due to the Bunsen burners. I enjoyed observing the color changes, and I look forward to hopefully doing more labs like this one!
Tuesday, February 16, 2016
Unit Exam...
I have heard countless stories of how the acid base unit test is THE most difficult test of the year, and I arrived to school last Thursday fearing for my life. Apparently a good chunk of this unit test was basic vocab, essentially textbook comprehension questions, which I'm pretty good at memorizing. I've heard there are maybe one or two math problems that are very hard, but other than that, the rest of the test went by in a breeze. I'm hopeful that this is the case because I would be very disappointed if I got a grade similar to the last exam.
Some links I used to study and will be using for the final:
http://www.chemtutor.com/acid.htm#math
https://www.khanacademy.org/science/chemistry/acid-base-equilibrium/titrations/v/titration-calculation-example
Some links I used to study and will be using for the final:
http://www.chemtutor.com/acid.htm#math
https://www.khanacademy.org/science/chemistry/acid-base-equilibrium/titrations/v/titration-calculation-example
Unknown Acid Lab!
Unknown Acid Lab
Last week, we worked on another titration lab that is pretty much identical to the previous titration lab (Acetic Acid lab). The only thing that's changed is that we're not using acetic acid, rather an unknown acid. We still titrated the unknown acid with NaOH, just like the acetic acid lab. Another thing that's been added to this lab is that we have to heat the solution as the unknown acid does not dissolve as easily as acetic acid. It's pretty neat seeing the solution turn into a mini tornado when the magnetic thingamajig is added to the flask and spins due to the magnet in the heater.One frustrating thing about this lab is that we can't seem to get a good percent error like last lab where we got less than 1% error! The first trial of this lab gave us a 14% error, which is definitely not preferred, and the second trial gave us a 7% error, however when we re-did the experiment the following day, we were able to get a less than 1% error like the first lab.
 |
| Solution being stirred |
Nuetralization Reactions?!
In Ahrrenius acid/base reaction the products are the same - a salt and water
- salt is formed from the cation of a base, and the anion of the acid
- the strengths of the parent acid and parent base are the determiners of the strength of the salt
- strong acid + strong base = neutral salt
- strong acid + weak base = acidic salt
- weak acid + strong base = basic salt
- weak acid + weak base = neutral salt
- the strongest parent runs the reaction
The Hydrogen Ion in Water
- the hydrogen ion is simply a proton (nucleus of a hydrogen atom without its valence electron)
- is water, clusters of hydrated hydrogen ions form
- the simplest cluster is hydronium (H3O)+
- H+ is the same is H3O+
Water as an Acid and a Base
Water can act as an acid because it can donate a proton. Water can be a base because it can accept a proton to form the hydronium ion.
1st Titration Lab
Titration Lab
Last week, we worked on a new lab. The purpose of the lab is to standardize a solution of NaOH with KHP, and then, use that solution's molarity to determine the percentage of acetic acid is in vinegar. To start, we filled the buret with NaOH, and measured out some KHP on a weigh boat. We transfered the KHP to a flask, and filled it with water to make a solution of 75 mL. Next, we added three drops of phenolphthalein to act as a color indicator for the endpoint. Lastly, we slowly drained the NaOH solution into the flask until the solution reached a permanent pink color. This was the hard part as we never knew exactly when to stop, but then again, we couldn't go too slowly as we needed to finish within a class period. Although, I thought I sometimes went too overboard with the NaOH, we ended up getting a 4.50% error! All in all, I enjoyed this lab and watching the clear liquid turn into a bright pink in a matter of drops.
 |
| KPH |
| The solution turns pink where the base hits it, but then soon disappears |
 |
| The endpoint has been reached! |
Acid-Base quiz
Acids & Bases Weekly Quiz
The first quiz proved to be quite a relief. All throughout the day before, I had been stressing over this quiz as I had not done so well on the last unit test. Thankfully, when it came down to actually taking it, it was pretty much half as hard as I thought it would be. I had no trouble answering any of the questions, and I had time to double (and for some, triple) check my answers. These are the kinds of quizzes I thank the chemistry gods for blessing upon me. I have a good feeling about this one.Hopefully, the rest of the unit won't get much harder than what we've learned so far, but I'm probably jinxing myself.
Here are some of the links I used to study with which may come in handy for the unit test:
https://www.youtube.com/watch?v=Xeuyc55LqiY
https://www.youtube.com/watch?v=l5fk7HPmo5g
http://www.chemteam.info/AcidBase/AcidBase-Problems&Video.html
The Acid-Base Lessons
Lessons on Acid-Base
We started our new unit on acids & bases on Wednesday 2 weeks ago. The majority of the lessons are not very complex nor difficult to understand. I may just need to study some of the vocab again. Wednesday's lecture consisted of identifying the differences between acids and bases, strong vs. weak, and Arrhenius vs Bronsted-Lowry acids. This was probably the most difficult out of the two lectures we've had so far as it was very vocab-packed, and there are some rules that need to be memorized in order to identify what type of acid or base a substance is. Recent lectures were more math based in that we had. It was basic math and involved a simple formula to find the M of either H or OH ions in a reaction. Overall, these two lessons have not been difficult, and I am hoping that the rest of the unit will not be super bad (but it probably will).A good summary of what we learned:http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text12/Tx121/tx121.html
Thursday, January 21, 2016
Murder Lab Day 2
Day Two consisted of weighing the dry filter paper that contained the solid from the reaction. The solid was a pea green powder.
After weighing the filter paper, we subtracted this mass from the mass of just the filter paper to figure out the mass of the solid. Then, we calculated the molarity of this substance and found out that it was roughly 0.07 M. To figure out the identity of the unknown substance, we knew that it must have been silver nitrate because when silver nitrate combines with sodium carbonate, it should make a solid, which is exactly what happened in lab. The 0.07 molarity matched Mr. Green's description, as he was found with traces of silver nitrate with a molarity between 0.05 and 0.15 M.
Murder Lab Day 1
The murder investigation lab seemed to be a very interesting lab. I love the idea of solving a a mystery while incorporating chemistry into it. It gives a taste of what forensic investigators do in reality. Today's task was combining an unknown solution with sodium carbonate in a beaker. Both reactants were clear, but when combined, the product was a milky white substance.
Then, we poured this substance into a funnel with filter paper inside an empty Erlenmeyer flask to collect any solid. While letting the liquid drip down into the flask, I noticed there was a sand-like substance forming inside the liquid, showing that the reaction definitely produced a solid. Thus, it can be inferred that the unknown substance is silver nitrate, due to the fact that it should produce a solid when reacted with sodium carbonate based on solubility rules.
Subscribe to:
Comments (Atom)












