
Monday, May 9, 2016
Airbag stuff...
I managed to pass the lab today, I know, what a surprise! I was super nervous for it, but I thankfully came through! I think everyone in my class passed the lab as well, so that's cool.


Probably got an A?
On Thursday, we took a quiz on gas laws. The quiz was not that bad at all, and I think I scored pretty well. The lectures we've learned so far were not too hard and only consisted of formulas. I am looking forward to our next lessons.
| http://www.gettyimages.com/detail/photo/whew-what-a-relief-royalty-free-image/471428407 |
Something with pvnrt
Today, we had another lecture for the gas laws unit. This was yet another formula lecture, one that was not difficult at all and mostly involved basic plug-n-chug. I am really nervous for the lab on Monday as it is pass-fail, and I don't 100% trust myself with being accurate in my calculations. Hopefully, it goes well.
Some review links:
https://www.khanacademy.org/science/chemistry/gases-and-kinetic-molecular-theory/ideal-gas-laws/v/ideal-gas-equation-pv-nrt
http://www.learnchem.net/practice/gases.shtml
http://www.mikeblaber.org/oldwine/chm1045/notes/Gases/IdealGas/Gases04.htm
| http://www.calctool.org/CALC/chem/c_thermo/ideal_gas |
Some review links:
https://www.khanacademy.org/science/chemistry/gases-and-kinetic-molecular-theory/ideal-gas-laws/v/ideal-gas-equation-pv-nrt
http://www.learnchem.net/practice/gases.shtml
http://www.mikeblaber.org/oldwine/chm1045/notes/Gases/IdealGas/Gases04.htm
Gas Lectures
The gas laws unit has proven to be pretty straightforward and simple. The first lectures we've had are centered on one major formula: the combined gas law formula. We've gone through various different problems concerning the gas law, only with different constants. Additionally, we've learned different units of pressure, which I believe is the only difficult part of this unit: memorizing all the different conversions and units.
Some links I've been using to study with for the quiz tomorrow:
| https://prezi.com/8ywewmiga8tq/combined-gas-law/ |
Some links I've been using to study with for the quiz tomorrow:
since when does it take 15 min to complete a test
The unit test for energy was a bit of a doozy. Since our schedule for the day was messed up, we were only given 28 minutes to complete the test. Seems impossible, right? It actually wasn't as bad as I thought it was going to be. There were only 20 questions, and I managed to finish with a couple minutes to spare. We got our grades back and I managed to score what I needed to keep my grade where it needs to be in order to exempt the final exam.
Studying for the final
| https://commons.wikimedia.org/wiki/File:Phase_changes.svg |
| http://sonoranweeklyreview.com/with-linn-energy-llc-nasdaqline-sandridge-energy-sdoc-and-midstates-petroleum-mpoy-depleting-their-revolving-credit-lines-banks-scramble/ |
Some links I used for studying:
http://www.aboutthemcat.org/chemistry/thermochemistry.php
https://www.chem.wisc.edu/deptfiles/genchem/netorial/rottosen/tutorial/modules/intermolecular_forces/02imf/imf2.htm
https://www.sophia.org/tutorials/thermochemistry-calculating-energy-of-phase-change
https://www.khanacademy.org/test-prep/mcat/chemical-processes/covalent-bonds/a/intramolecular-and-intermolecular-forces
Metal, Heat, some other stuff...
Yesterday, we did a lab to measure the specific heats of copper and lead by heating the metals in a hot water bath and figuring out the variables for MCAT. I'm pretty satisfied because our copper trial yielded only a 4% error for the specific heat!
Some review links for the first lesson:
Boat Races...
Last week, we conducted our boat races. I'm proud to say that my partner and I won last place. It was a really fun experience, and it definitely was a relief from the usual lecture/test stress. We made it out of an eggnog carton...

A real boat that runs on biodiesel: https://www.youtube.com/watch?v=4EggCi3qM2A
So, The biodiesel worked lol
On Friday, we found our biodiesel separated into three layers! The top and bottom layers were impurities, and our actual biodiesel was the middle layer. To test if our biodiesel worked, we filled an empty tea light wick with the biodiesel and lit it. Sure enough, the flame was sustained for quite a bit, indicating that our biodiesel worked great!
Biodiesel, I think?
Here's some research I accumulated regarding the topic of Biodiesel:
General Applications
Benefits;
General Applications
- Biodiesel can be used in pure form (B100) or may be blended with petroleum diesel at any concentration in most injection pump diesel engines
- With 80-90% of oil spill costs invested in shoreline cleanup, there is a search for more efficient and cost-effective methods to extract oil spills from the shorelines. Biodiesel has displayed its capacity to significantly dissolve crude oil, depending on the source of the fatty acids
- Depending on the engine, this might include high pressure injection pumps, pump injectors (also called unit injectors) and fuel injectors
Benefits;
- domestically produced, clean-burning, renewable substitute for petroleum diesel. Using biodiesel as a vehicle fuel increases energy security, improves air quality and the environment, and provides safety benefits
- Biodiesel is produced in the U.S. and used in conventional diesel engines, directly substituting for or extending supplies of traditional petroleum diesel-- no need for petroleum imports
- reduces nitrogen oxide (NOx) emissions to near zero levels
- reduces greenhouse gas emissions because carbon dioxide released from biodiesel combustion is offset by the carbon dioxide absorbed while growing the soybeans or other feedstock
- B100 use reduces carbon dioxide emissions by more than 75% compared with petroleum diesel. Using B20 reduces carbon dioxide emissions by 15%
- Biodiesel improves fuel lubricity and raises the cetane number of the fuel
- Diesel engines depend on the lubricity of the fuel to keep moving parts from wearing
How to make biodiesel
On Thursday, we began the process of making our own biodiesel! First, we took recycled peanut oil and combined it with a mixture of KOH and methanol. Then, we put a flask of this mixture into a hot water bath and boiled it for about 20 minutes. Lastly, we let it cool and settle, upon which it began to separate into two layers.
Make your own biodiesel at home!: http://www.make-biodiesel.org/
 |
| Hot water bath |
 |
| Viscosity test |
Make your own biodiesel at home!: http://www.make-biodiesel.org/
How it's made: https://www.youtube.com/watch?v=xLa83KIaEyw
Bio Diesel Video
This past week, we've been working on a video project to submit to the National Lung Association biodiesel contest. Our video explained the benefits and debunked myths about biodiesel. We made a stop-motion video which took quite a while as we had to take multiple pictures to create a full-length animated video. However, it was definitely worth it as our final product was really cool! I'm really looking forward to checking out other students' videos.
Unit Exam, not too bad
Our unit exam for the chemical bonding unit was yesterday. Initially, I was nervous about the content of the unit as a whole, it was a little tricky to study and remember, but I received a perfectly adequate grade. It was Lewis structure heavy, along with some emphasis on electronic geometry & molecular shape. Here are some fun pictures pertaining to our unit.


More lectures...
Chemical Bonding Lectures
Our current unit is based on a single concept: a (covalent) bond is formed by sharing electrons. Atoms are constantly in search of electrons to fill their valence shells, which will make them stable. More often than not, it's 8 electrons except for Be, B, H, & He. We learned lewis structures, formal charge, electron geometry, and much more involving bonding.
Modeling Activity
Last week, we went to the library for a lab. It was interesting as it was in the library where we drew on the tables and played around with building pieces to create Lewis structures (felt just like kindergarden again). I enjoyed this lab as it allowed me to practice Lewis strucutres, something I am still quite uneasy about. It was also great to have Mrs. Frankenberg hovering around, available to answer any questions (which I had a lot of). I will definitely be finishing the rest of the lab on my own for additional practice.
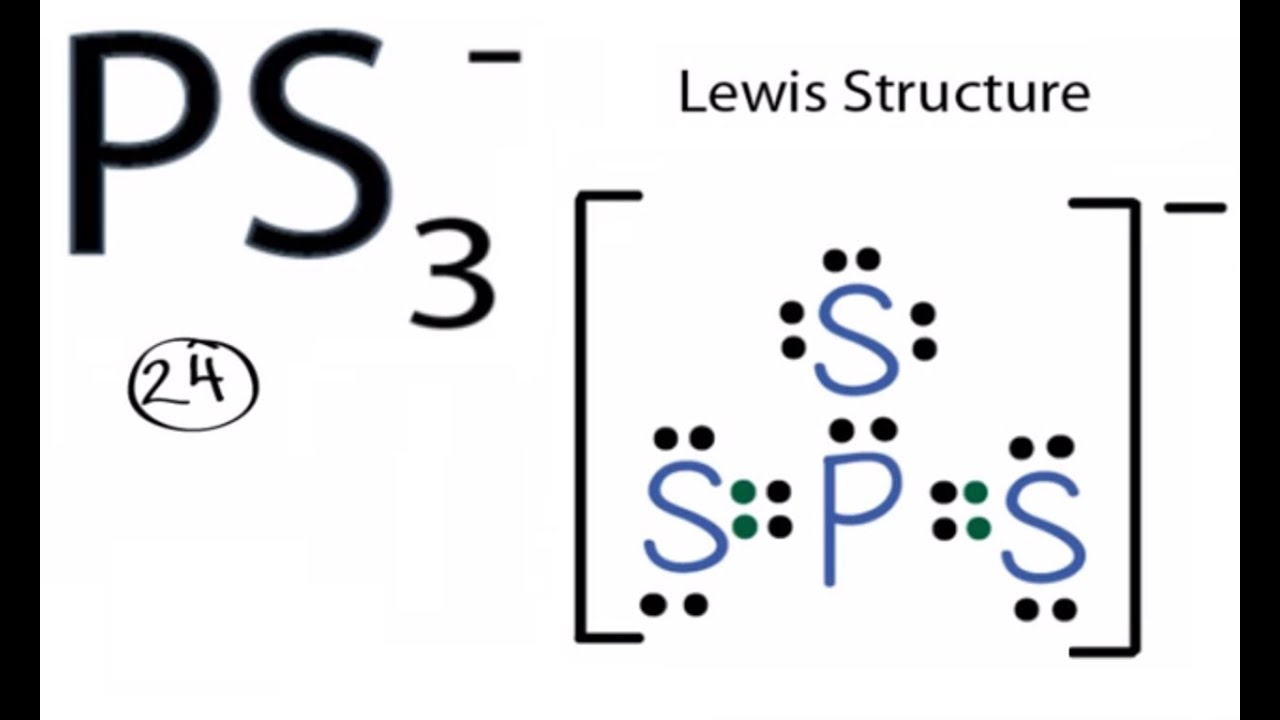
Some more practice: http://web.gccaz.edu/~ksmith8/chm151_files/Lewispractice.pdf
The first day...
Lecture Reflections
We've started the chemical bonding unit, and it's been a bit of a rollercoaster. At first, the concepts were fairly easy to understand, but as the lectures progress, the confusion keeps piling on. I do not believe this unit will be too difficult; it's just the fact that there are SO many things to remember to do problems correctly. I think with a lot of practice and reviewing this unit will not be too bad.
Some practice:
| http://www.slideshare.net/Drahmadfahmi/04-chemical-bonds |
| http://www.roymech.co.uk/Related/Chemistry/Lewis_dot_structure.html |
Some practice:
Subscribe to:
Comments (Atom)





